A Spectral Mystery – Solved!
We were recently contacted by a university Safety Officer (SO, from now on). A researcher using our Model SFA Stereo Microscope Fluorescence Adapter with Royal Blue excitation thought that the light source might also be emitting hazardous shortwave ultraviolet radiation. Properly concerned for general lab safety he stopped using the system until he could confirm that there was no UV hazard. Our immediate reaction was ‘Of course that’s not possible!’, but we take any safety concerns very seriously and worked with the SO to find a way to clearly confirm our belief. The whole process was instructive and we wanted to share it here. We’ll tell you why the researcher thought that there might be UV present, and what we did to confirm that there was not.
Why the researcher thought there was shortwave ultraviolet radiation
The researcher was working with ethidium bromide (EtBr), a nucleic acid fluorescent stain widely used in molecular biology for techniques such as agarose gel electrophoresis. His understanding was that EtBr required excitation in the range of 300nm, well down into the ultraviolet, so when he placed a gel under the SFA and saw some EtBr fluorescence he concluded that there must be 300nm present.
It is not hard to understand why he thought that EtBr required shortwave UV. UV transilluminators (for example, those from VWR, Spectroline, or UVP) are routinely sold for this purpose. The Wikipedia entry on EtBr states:
“Detection typically involves a gel containing nucleic acids placed on or under a UV lamp. Since ultraviolet light is harmful to eyes and skin, gels stained with ethidium bromide are usually viewed indirectly using an enclosed camera, with the fluorescent images recorded as photographs. Where direct viewing is needed, the viewer’s eyes and exposed skin should be protected.”
There is also literature, both scientific and from manufacturers, that describes the use of these short wavelengths for EtBr fluorescence – Biotechniques article, Analytical Chemistry article, ThermoFisher product page.
Based on the information above, it is easy to understand why one would have a strong association between ultraviolet and EtBr fluorescence. With this mindset the researcher was absolutely correct to be concerned when he saw fluorescence under our blue light source.
Why we knew there was no shortwave ultraviolet radiation
As soon as we were asked the question by the SO we knew that something else must be going on. There are some very good technical reasons why we knew there would be no UV present.
1 – Our light sources utilize LEDs (light emitting diodes). LEDs are semiconductors engineered to have specific band gaps. The wavelength output of the LED is determined by the energy associated with the band gap. In plain English this means that an LED emits a relatively narrow band of wavelengths centered around the design wavelength. An LED emitting in the mid-blue (440 – 460nm) like our Royal Blue light heads will simply not be a source of radiation in the 300nm range. [And if it were, the manufacturers of the LED would be sure to provide strong warnings (which they don’t).]
2 – The focusing optics in the Royal Blue light head do not transmit ultraviolet light. In order to get a tight beam for strong excitation under the stereo microscope we include a focusing element in the light head. This is an acrylic optic that is transparent at visible wavelengths but opaque in the ultraviolet. [We do offer an ultraviolet light head for the SFA (longwave UV, 360-380nm range) and part of the added expense of that particular source is the special UV-transmitting focusing optics.]
The mystery
So we have a mystery: if the Royal Blue light head does not emit shortwave ultraviolet, and if shortwave ultraviolet is needed to excite EtBr, then why is there fluorescence? Half of the statement above has to be incorrect, and it is the second half. We knew there was no shortwave UV, but just saying so was not enough – the researcher saw the light exciting EtBr fluorescence. We needed evidence and practical tests.
Evidence that you do not need shortwave ultraviolet to see EtBr
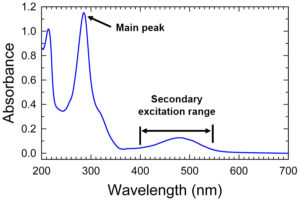
Here is the excitation spectrum for EtBr:
(Click image for larger view)
[By Mark Somoza – Own work, CC BY 2.5]
An excitation spectrum is essentially a graph of the relative ability of different wavelengths of light to produce fluorescence in a subject. The graph shows that shortwave UV, around 300nm, is by far the most efficient wavelength range for exciting EtBr fluorescence. But it also teaches that it is not the only wavelength range for this purpose. The broad, low secondary peak extending from about 400 to 570nm encompasses wavelengths that can also excite EtBr fluorescence, albeit much less efficiently than the short UV wavelengths.
There is a lot of additional support for this. If you Google ‘ethidium bromide excitation emission’ you will find many links to tabulations of excitation and emission peaks for a variety of fluorophores. Wavelengths recommended for EtBr excitation in these tables include 493, 510, 518, 524, and 545nm!
In addition, there are companies that sell blue-light transilluminators that can be used for EtBr. Check out this blog post from Vernier.
Visual testing
I sent this information to the SO, but together we wanted to also provide hard evidence to the researcher. The SO acquired two optical filters:
- UV-only – transmits ultraviolet (<400nm) but no visible light
- VIS-only – transmits visible light (>400nm) but no ultraviolet
He also acquired a target with components that fluoresced under ultraviolet and visible light and performed the following tests with the researcher:
- UV light source or RB light source, no filter
- Result – fluorescence visible
- Conclusion – demonstrates that both light sources can excite the fluorescence
- UV light source, UV-only filter
- Result – fluorescence visible
- Conclusion – confirms that the UV-only filter will transmit UV if it is present
- UV light source, VIS-only filter
- Result – no fluorescence
- Conclusion – the VIS-only filter is an effective UV blocker
- SFA Royal Blue (RB) source, UV-only filter
- Result – no fluorescence
- Conclusion – there is no UV output from the RB light
- SFA Royal Blue (RB) source, VIS-only filter
- Result – fluorescence visible
- Conclusion – visible blue light can excite the fluorescence
This documentation and testing finally proved what needed to be proved:
- You don’t necessarily need ultraviolet light to excite EtBr fluorescence
- The RB light source does not emit ultraviolet light
Conclusion
Working collaboratively with the university SO we demonstrated to the researcher’s satisfaction that there was no dangerous UV radiation emitted by the Royal Blue light head. This was a valuable exercise in tying together underlying information about technology (LEDs), data on excitation spectra ranges, and simple, practical tests to understand why a phenomenon was happening.
Case closed!